Theoretical Framework:
The Conceptual Age, TPACK, and Content Area Literacy
The Conceptual Age
“Now, as the forces of Abundance, Asia, and Automation deeper and intensify, the curtain is rising on…the Conceptual Age. The main characters now are the creator and the empathizer, whose distinctive ability is mastery of R-Directed Thinking. …L-Directed Thinking remains indispensable. It’s just no longer sufficient. In the Conceptual Age, what we need instead is a whole new mind. …[W]e will need to complement our L-Directed reasoning by mastering six essential R-Directed aptitudes. …Design. Story. Symphony. Empathy. Play. Meaning. These six senses increasingly will guide our lives and shape our world” (Pink 2006).
“We are in the midst of another revolution that at least rivals the Industrial Revolution. This revolution…is significantly changing our society and thus the value of knowledge and talents. We must then ask the same question: What should schools teach in order to prepare our children for the global and digital economy? …In the new era, we need more diverse talents rather than standardized laborers, more creative individuals rather than homogenized test takers, and more entrepreneurs rather than obedient employees. Furthermore, this new era requires all citizens to think globally, to understand other cultures, to have the ability to interact with others, and to competently handle changes and complexity. …American education needs major changes, but the changes should be oriented to the future instead of the past or present. The changes should be made out of hope for a better tomorrow instead of fear of losing yesterday or today. And as such, changes, I suggest, should include expanding the definition of success, personalizing education, and viewing schools as global enterprises.” (Zhao 2009).
“[M]y contention is, all kids have tremendous talents. And we squander them, pretty ruthlessly. …My contention is that creativity now is as important in education as literacy, and we should treat it with the same status. …“[K]ids will take a chance. If they don't know, they'll have a go. Am I right? They're not frightened of being wrong. Now, I don't mean to say that being wrong is the same thing as being creative. What we do know is, if you're not prepared to be wrong, you'll never come up with anything original -- if you're not prepared to be wrong. And by the time they get to be adults, most kids have lost that capacity. They have become frightened of being wrong. …We stigmatize mistakes. And we're now running national education systems where mistakes are the worst thing you can make. And the result is that we are educating people out of their creative capacities. Picasso once said this -- he said that all children are born artists. The problem is to remain an artist as we grow up. I believe this passionately, that we don't grow into creativity, we grow out of it. Or rather, we get educated out if it. …And the consequence is that many highly talented, brilliant, creative people think they're not, because the thing they were good at [in] school wasn't valued, or was actually stigmatized. And I think we can't afford to go on that way.” – Sir Ken Robinson, 2006
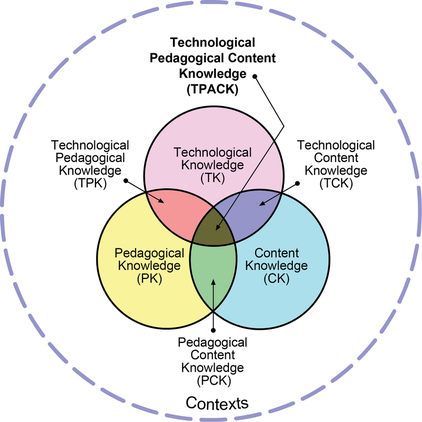
TPACK: Technological Pedagogical Content Knowledge
"The advent of digital technology has dramatically changed routines and practices in most arenas of human work. Advocates of technology in education often envisage similar dramatic changes in the process of teaching and learning. …However, the relationships between content (the actual subject matter that is to be learned and taught), pedagogy (the process and practice or methods of teaching and learning), and technology (both commonplace, like chalkboards, and advanced, such as digital computers) are complex and nuanced. Technologies often come with their own imperatives that constrain the content that has to be covered and the nature of possible representations. These decisions have a ripple effect by defining, or in other ways, constraining, instructional moves and other pedagogical decisions. So it may be inappropriate to see knowledge of technology as being isolated from knowledge of pedagogy and content. …[The TPACK] framework emphasizes the connections, interactions, affordances, and constraints between and among content, pedagogy, and technology. In this model, knowledge about content (C), pedagogy (P), and technology (T) is central for developing good teaching. However, rather than treating these as separate bodies of knowledge, this model additionally emphasizes the complex interplay of these three bodies of knowledge” (Mishra and Koehler 2006).
“Now, as the forces of Abundance, Asia, and Automation deeper and intensify, the curtain is rising on…the Conceptual Age. The main characters now are the creator and the empathizer, whose distinctive ability is mastery of R-Directed Thinking. …L-Directed Thinking remains indispensable. It’s just no longer sufficient. In the Conceptual Age, what we need instead is a whole new mind. …[W]e will need to complement our L-Directed reasoning by mastering six essential R-Directed aptitudes. …Design. Story. Symphony. Empathy. Play. Meaning. These six senses increasingly will guide our lives and shape our world” (Pink 2006).
“We are in the midst of another revolution that at least rivals the Industrial Revolution. This revolution…is significantly changing our society and thus the value of knowledge and talents. We must then ask the same question: What should schools teach in order to prepare our children for the global and digital economy? …In the new era, we need more diverse talents rather than standardized laborers, more creative individuals rather than homogenized test takers, and more entrepreneurs rather than obedient employees. Furthermore, this new era requires all citizens to think globally, to understand other cultures, to have the ability to interact with others, and to competently handle changes and complexity. …American education needs major changes, but the changes should be oriented to the future instead of the past or present. The changes should be made out of hope for a better tomorrow instead of fear of losing yesterday or today. And as such, changes, I suggest, should include expanding the definition of success, personalizing education, and viewing schools as global enterprises.” (Zhao 2009).
“[M]y contention is, all kids have tremendous talents. And we squander them, pretty ruthlessly. …My contention is that creativity now is as important in education as literacy, and we should treat it with the same status. …“[K]ids will take a chance. If they don't know, they'll have a go. Am I right? They're not frightened of being wrong. Now, I don't mean to say that being wrong is the same thing as being creative. What we do know is, if you're not prepared to be wrong, you'll never come up with anything original -- if you're not prepared to be wrong. And by the time they get to be adults, most kids have lost that capacity. They have become frightened of being wrong. …We stigmatize mistakes. And we're now running national education systems where mistakes are the worst thing you can make. And the result is that we are educating people out of their creative capacities. Picasso once said this -- he said that all children are born artists. The problem is to remain an artist as we grow up. I believe this passionately, that we don't grow into creativity, we grow out of it. Or rather, we get educated out if it. …And the consequence is that many highly talented, brilliant, creative people think they're not, because the thing they were good at [in] school wasn't valued, or was actually stigmatized. And I think we can't afford to go on that way.” – Sir Ken Robinson, 2006
TPACK: Technological Pedagogical Content Knowledge
"The advent of digital technology has dramatically changed routines and practices in most arenas of human work. Advocates of technology in education often envisage similar dramatic changes in the process of teaching and learning. …However, the relationships between content (the actual subject matter that is to be learned and taught), pedagogy (the process and practice or methods of teaching and learning), and technology (both commonplace, like chalkboards, and advanced, such as digital computers) are complex and nuanced. Technologies often come with their own imperatives that constrain the content that has to be covered and the nature of possible representations. These decisions have a ripple effect by defining, or in other ways, constraining, instructional moves and other pedagogical decisions. So it may be inappropriate to see knowledge of technology as being isolated from knowledge of pedagogy and content. …[The TPACK] framework emphasizes the connections, interactions, affordances, and constraints between and among content, pedagogy, and technology. In this model, knowledge about content (C), pedagogy (P), and technology (T) is central for developing good teaching. However, rather than treating these as separate bodies of knowledge, this model additionally emphasizes the complex interplay of these three bodies of knowledge” (Mishra and Koehler 2006).
Content Area Literacy
“To become literate in a content area, students must understand how to use reading, writing, talking, and viewing to learn. Integrating these thinking/learning processes helps students to better understand what they are reading about, writing about, talking about in classroom discussion, or viewing on a computer screen or video monitor. …Texts include not only print forms of communication but also nonprint forms that are digital, aural, or visual in nature. Texts in content area classrooms represent sets of potential meanings and signifying practices, whether the text is a novel in an English class, the instructional conversation that takes place about the novel, or the made-for-television movie based on the novel. Helping students to think and learn with all kinds of text is an important responsibility of the content area teacher. Johannes Gutenberg’s invention of movable type in the fifteenth century resulted in a revolution of ideas. Printed texts in the hands of the masses changed the face of literacy and learning in much the same way that multimodal information and communication technologies (ICT) are creating new literacies and ways of learning” (Vacca, Vacca, and Mraz 2011).
Pivotal Texts, Resources, and Coursework
The foundation upon which my understanding of what we
have come to call “new literacies” and “21st century skills” has
been built, I owe to Vacca and Vacca’s (2008, 2012) expanded definition of literacy from
their ubiquitous textbook, Content Area
Reading: Literacy and Learning Across the Curriculum. Before entering the classroom as a first-year
teacher, I was able to participate in a graduate course on Content Area Reading
(ECI 541) taught by Dr. Lois Huffman. As
the son of a school reading specialist, I had been taught that literacy was
important, and I perceived that the shifting energies of educational policy
were soon to make a strong lean toward targeted development of students’
literacy skills. Since my undergraduate
teacher preparation program for High School science had not included any sort
of focus on these skills, I was thankful for the chance to learn. However, as a reluctant reader myself, I had
no way of connecting with students’ literacy needs – I was just as lost as they
were! When Dr. Huffman led me to
recognize Vacca and Vacca’s explanation of literacy as a multifaceted array of
skills for interacting with a huge range of information resources (texts), for
the first time, I felt that I, too, could be a reader.
For my Content Area Reading course, I was asked to build a personal "Reading Autobiography."
While my story includes many frustrating deterrents away from reading, I have also had several
important influences toward reading in my life. My grandfather and mother have always been
avid readers, and nonprint texts like music lyrics have always had a strong impact on my
critical thinking related to media.
While my story includes many frustrating deterrents away from reading, I have also had several
important influences toward reading in my life. My grandfather and mother have always been
avid readers, and nonprint texts like music lyrics have always had a strong impact on my
critical thinking related to media.
As I dove into reading myself, for the first time in my life, I discovered how much I had been missing all of these years. I had always loved books, but I had always hated reading… After sifting through my own history as a learner and as a reader, I was able to identify all of the “texts” that I “read.” According to Vacca and Vacca, I was always reading! I was an excellent reader! But I had always been told by teachers that I was a poor, slow reader. The problem with all of this was that I had been forced to follow a definition of literacy – exclusively reading – and a definition of text – exclusively print resources – that I did not identify with. Finally, as a pre-service teacher, I was empowered to change that for myself.
By understanding literacy as not only reading and writing, but as reading and writing and speaking and listening and viewing and representing (Dr. Huffman’s addition for Vacca and Vacca), and by learning that, especially in the new learning ecology (Spires et al 2012) of the digital world, a text does not have to be printed words on a white page, but could be any one of a massive variety of resources and tools for learning, I harnessed reading (and all of its accompanying literacy modes) for myself, and for my students.
In Dr. Hiller Spires’ course in New Literacies and Media (ECI 546), I was able to apply my newly discovered love of reading to two books that captivated my interest as a beginning teacher, and jumpstarted my graduate learning in the NLGL program at NCSU. Still to this day, Daniel Pink’s (2006) A Whole New Mind, and Yong Zhao’s (2009) Catching Up or Leading the Way serve as pillars of the development of my educational philosophy. Together, these texts shaped my beliefs about the past, present, and future of schooling in the United States, and have provided for me an intellectual basis for making decisions to preserve opportunities for creativity and innovation in my classroom – some of the same opportunities that I do not feel were afforded me when I was a student, contributing to my own academic struggles. Even further, these books have shaped my understanding of my students. When I find my students experiencing academic struggles, I think about the strengths they offer – the Conceptual Age senses they bring to our classroom – that I can harness to help them experience success. These resources served as some of the original building blocks that have helped me become the educator that I am today, and the educational leader I aspire to become.
Theory to Practice: "21st Century Literacies" in Action
Combining chemical bonding,
molecular structure, and neurochemistry to address the risks of teen drug abuse
In a self-paced, project-based unit for Chemistry, students inquire into the nature of chemical bonding and three-dimensional molecular structures as they explore the risks, representations, contributing factors, and biochemical mechanisms of drug use and abuse by adolescents in American society. The unit seeks to provide a rigorous, meaningful, high-interest context for an often difficult aspect of the Chemistry curriculum, while addressing concerns about underage drinking and increased abuse of prescription medical drugs among students in our school’s local community.
The unit begins with a classwide, teacher-led discussion based on the reading of a news article, students’ personal reflections and experiences, and the viewing of a documentary about teens’ questions and interests in the use of a range of illegal substances. After building shared, background knowledge, as well as a sense of safety and openness, students initiate a teacher-designed series of resource-based activities – readings, illustrative videos, interactive digital games, and inquiry-based labs – to explore and learn about the chemistry of the human brain, drug substance’s biochemical affects on the human brain, and how these are explained by molecular structure and the underlying principles of chemical bonding. Students engage with complex graphics, concepts, and vocabulary within the context of a variety of text resources. Because the unit is self-paced, teachers are available to provide support where needed and to respond to extension questions from students as they arise. Teachers provide materials for all lab activities as a method of monitoring student progress through assigned activities, and students complete short, formal formative assessments on an ongoing basis to ensure that they are actually completing assignments, experiencing success on an independent basis, and mastering the unit’s Chemistry content standards.
At the end of the unit, students complete two summative assessments, including a formal written test with an open-ended essay response and a personal project that allows for individual choice and creativity. The project is permitted to take one of several forms, all of which lead the student to connect their understanding of the Chemistry content to the significance of the teen drug abuse context. Some students complete and document action research surveys to examine attitudes and awareness among their peers while others design digital infographics to share what they have learned about a specific drug substance. Still others choose to complete a formal research essay while some students build an interactive, three-dimensional model of a specific drug molecule, including explanation of its molecular structure and biological impact.
Through 21st century modes of teaching, learning, responding, and assessing, in the context of controversial, complex social issues directly related to the lived experiences of students, this project-based inquiry unit demonstrates the sort of learning that is both rigorous and consequential for today’s students. With support for activating background knowledge, building science content vocabulary through text and graphics, and by providing input through a variety of multimodal resources, the unit is built on a foundation of the principles of content-area literacy.
In a self-paced, project-based unit for Chemistry, students inquire into the nature of chemical bonding and three-dimensional molecular structures as they explore the risks, representations, contributing factors, and biochemical mechanisms of drug use and abuse by adolescents in American society. The unit seeks to provide a rigorous, meaningful, high-interest context for an often difficult aspect of the Chemistry curriculum, while addressing concerns about underage drinking and increased abuse of prescription medical drugs among students in our school’s local community.
The unit begins with a classwide, teacher-led discussion based on the reading of a news article, students’ personal reflections and experiences, and the viewing of a documentary about teens’ questions and interests in the use of a range of illegal substances. After building shared, background knowledge, as well as a sense of safety and openness, students initiate a teacher-designed series of resource-based activities – readings, illustrative videos, interactive digital games, and inquiry-based labs – to explore and learn about the chemistry of the human brain, drug substance’s biochemical affects on the human brain, and how these are explained by molecular structure and the underlying principles of chemical bonding. Students engage with complex graphics, concepts, and vocabulary within the context of a variety of text resources. Because the unit is self-paced, teachers are available to provide support where needed and to respond to extension questions from students as they arise. Teachers provide materials for all lab activities as a method of monitoring student progress through assigned activities, and students complete short, formal formative assessments on an ongoing basis to ensure that they are actually completing assignments, experiencing success on an independent basis, and mastering the unit’s Chemistry content standards.
At the end of the unit, students complete two summative assessments, including a formal written test with an open-ended essay response and a personal project that allows for individual choice and creativity. The project is permitted to take one of several forms, all of which lead the student to connect their understanding of the Chemistry content to the significance of the teen drug abuse context. Some students complete and document action research surveys to examine attitudes and awareness among their peers while others design digital infographics to share what they have learned about a specific drug substance. Still others choose to complete a formal research essay while some students build an interactive, three-dimensional model of a specific drug molecule, including explanation of its molecular structure and biological impact.
Through 21st century modes of teaching, learning, responding, and assessing, in the context of controversial, complex social issues directly related to the lived experiences of students, this project-based inquiry unit demonstrates the sort of learning that is both rigorous and consequential for today’s students. With support for activating background knowledge, building science content vocabulary through text and graphics, and by providing input through a variety of multimodal resources, the unit is built on a foundation of the principles of content-area literacy.
|
|
Students begin by building background knowledge and interest through class discussion, viewing a documentary, and reading a short introductory article provided by the teacher.
Students then begin exploring the chemistry of chemical bonding, including types of bonding, and two- and three-dimensional representations of simple and complex molecules through physical and digital models. Students are led to connect this with the human brain's neurochemical receptors, recognizing that the three-dimensional shapes of molecules are the key to how they affect our bodies. Students then independently inquire into the biochemical impact of specific medical and illicit drug molecules using interactive tools from the University of Utah's Learn.Genetics program.
|
|

Framing science as a global, historical endeavor, through the
molecules that built the world
In June of 1812, Napoleon Bonaparte led almost a half million French soldiers into Russia beginning what he called the Second Polish War. After pushing the Russian army further into its own territories over three months of small victories, the French army was forced to chase the Russians through the country until they were weak in numbers, rations, and munitions. As the Russian winter took hold, the French army, still outnumbering and outmaneuvering the Russians, was forced to concede and retreat, not due to a lack of military might, but due to their inability to ward off the harsh cold. While they had been equipped by the French government with heavy coats, the buttons which held the coats closed had deteriorated away. This was because the small metal buttons were made of tin – an element known to suffer drastic, immediate weathering at temperatures common in the winter conditions. Napoleon’s retreat from Russia in November of 1812 is considered by many historians to be the defining moment in his eventual downfall and loss of power. What would have changed if the buttons had been made of a different metal, instead of tin? What if the French soldiers’ coats had been more reliable? How different would history have been? (LeCouter and Burreson 2003).
While this legend of tin buttons and the downfall of Napoleon Bonaparte have been largely debunked by chemists and historians, it does provoke an intriguing question: How have the simplest materials – metals, molecules, chemical substances – shaped the development, history, and circumstances of the world we live in today? Penny LeCouter and Jay Burreson’s (2004) book Napoleon’s Buttons: How 17 Molecules Changed the World explores this question by naming a series of chemical substances – vitamin C, salt, nylon, aspirin – and examining their roles as historical giants. I used LeCouter and Burreson’s book with my Chemistry students to explore history as a story of substances rather than a timeline of events. Assigning chapters of the book to small groups, students were led through a series of text analysis activities designed to create a rich discussion of their Chemistry learning over the course of the class, their awareness of national and global history, and their ability to apply and analyze a complex nonfiction text.
In this classwide project, students began by completing a close reading of their assigned chapters, following highlighting, annotation, and note-taking protocols. Students then completed a guided, written reflection designed to help them connect their understanding of the text to their understanding of related Chemistry concepts. Students then worked in groups to create a large concept map, diagramming the information from their assigned chapters in a dynamic, visual format. To complete the project, students wrote individual summaries of the text they read, posted these to a class wiki, and added graphics, images, and hyperlinks to resources that helped support the information they were communicating. Click below to view a few of the students' hypertext summaries:
molecules that built the world
In June of 1812, Napoleon Bonaparte led almost a half million French soldiers into Russia beginning what he called the Second Polish War. After pushing the Russian army further into its own territories over three months of small victories, the French army was forced to chase the Russians through the country until they were weak in numbers, rations, and munitions. As the Russian winter took hold, the French army, still outnumbering and outmaneuvering the Russians, was forced to concede and retreat, not due to a lack of military might, but due to their inability to ward off the harsh cold. While they had been equipped by the French government with heavy coats, the buttons which held the coats closed had deteriorated away. This was because the small metal buttons were made of tin – an element known to suffer drastic, immediate weathering at temperatures common in the winter conditions. Napoleon’s retreat from Russia in November of 1812 is considered by many historians to be the defining moment in his eventual downfall and loss of power. What would have changed if the buttons had been made of a different metal, instead of tin? What if the French soldiers’ coats had been more reliable? How different would history have been? (LeCouter and Burreson 2003).
While this legend of tin buttons and the downfall of Napoleon Bonaparte have been largely debunked by chemists and historians, it does provoke an intriguing question: How have the simplest materials – metals, molecules, chemical substances – shaped the development, history, and circumstances of the world we live in today? Penny LeCouter and Jay Burreson’s (2004) book Napoleon’s Buttons: How 17 Molecules Changed the World explores this question by naming a series of chemical substances – vitamin C, salt, nylon, aspirin – and examining their roles as historical giants. I used LeCouter and Burreson’s book with my Chemistry students to explore history as a story of substances rather than a timeline of events. Assigning chapters of the book to small groups, students were led through a series of text analysis activities designed to create a rich discussion of their Chemistry learning over the course of the class, their awareness of national and global history, and their ability to apply and analyze a complex nonfiction text.
In this classwide project, students began by completing a close reading of their assigned chapters, following highlighting, annotation, and note-taking protocols. Students then completed a guided, written reflection designed to help them connect their understanding of the text to their understanding of related Chemistry concepts. Students then worked in groups to create a large concept map, diagramming the information from their assigned chapters in a dynamic, visual format. To complete the project, students wrote individual summaries of the text they read, posted these to a class wiki, and added graphics, images, and hyperlinks to resources that helped support the information they were communicating. Click below to view a few of the students' hypertext summaries:
Zoe's summary of "Pepper, Nutmeg, and Cloves" and the spicey molecules of the Age of Discovery.
Nadia's summary of "Isoprene" - the key molecule to the creation of rubber.
Justin's summary of "Ascorbic Acid," more commonly known as vitamin C.
Nadia's summary of "Isoprene" - the key molecule to the creation of rubber.
Justin's summary of "Ascorbic Acid," more commonly known as vitamin C.
These hypertext summaries became the basis of a subsequent class project in which the class worked all together to build a massive timeline of the history of the world, through the eyes of Chemistry.